Tracing Tumor Evolution and Metastasis Using a Molecular Flight Recorder
In the opening plenary of the virtual AACR conference, Dr. Jonathan S. Weissman from the Whitehead Institute introduced the novel ‘molecular flight recorder’ technology that can record the history of tumor progression.
Phylogenetic trees are widely used in biology to peer into the evolution of a cell and help determine how two individual cells are close or distantly related to each other. Earlier, phylogenetic trees have been reconstructed from naturally occurring human tumors allowing researchers to address questions like how does a cancer initiate, metastasize or evade therapies?
Recording Tumor Progression in Real-Time
However, it is impossible to monitor the exact moment of metastasis as it is a transient event. Add to that, the technical limitations in sampling or capabilities in sequencing an adequate number of cells can diminish the depth of phylogenetic trees. Besides, this tool relies on the naturally occurring mutations or markers in a cancer cell lineage to estimate the relationship between cells. The molecular flight recorder technology overcomes these issues by recording tumor progression in real-time and also enabling the visualization of tumor metastasis as it happens.
Researchers use engineered human cancer cells where new heritable marks are introduced in the DNA using CRISPR/Cas9. These marks are inherited by daughter cells and accrue further changes during subsequent cell divisions. After the experiment, single-cell RNA sequencing (scRNA-seq) is used to measure the transcriptional state of the cell and the history of the tumor by reading the heritable marks from the transcribed RNA. This way, a rich, in-depth phylogenetic tree can be reconstructed from the output.
Dr. Weissman then talked about two collaborative studies where this technology was applied to follow tumor metastasis and trace tumor initiation and progression.
 Dr. Jonathan S. Weissman, Whitehead Institute, MIT
Dr. Jonathan S. Weissman, Whitehead Institute, MIT
Interesting Findings
In one study, the engineered cancer cells were injected into the mice lung (lung cancer xenograft mouse model) and were allowed to grow for a period of time. They were also able to visualize the metastasis in low resolution by imaging techniques. After the experiment, the RNA obtained from the different tissues of the sacrificed mice was subjected to scRNA-seq readout.
1. Unexpected Tumor Heterogeneity
The study revealed some very interesting phenomena. Unexpected heterogeneity was observed in metastatic states, meaning individual tumors were very different from each other. While some were weakly or highly metastatic, some others were non-metastatic. Further experiments showed that differences were not random and in fact when the cells of the same population were introduced in two different mice, they metastasized at a similar rate.
Interestingly, there were examples of large tumors with low metastatic potential and smaller tumors with high metastatic potential.
2. Distinct Transcriptional Programs Underlie Different Metastatic States
The study was also able to identify distinct genes that associated with either a higher or lower metastatic potential. They found that the gene KRT17 (that codes for Keratin 17 protein) was strongly expressed in low metastatic tumors than in highly metastatic tumors. Altering the Keratin 17 levels in vitro was sufficient to change the invasive properties of the tumor cells. This suggests there are several genes that can individually contribute to tumor invasiveness.
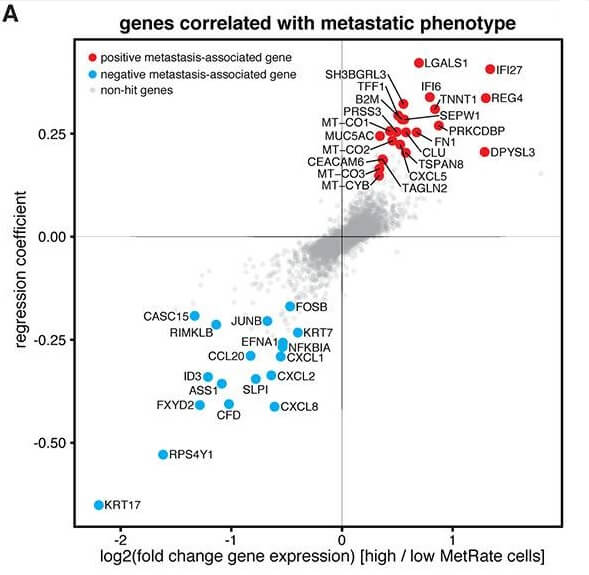
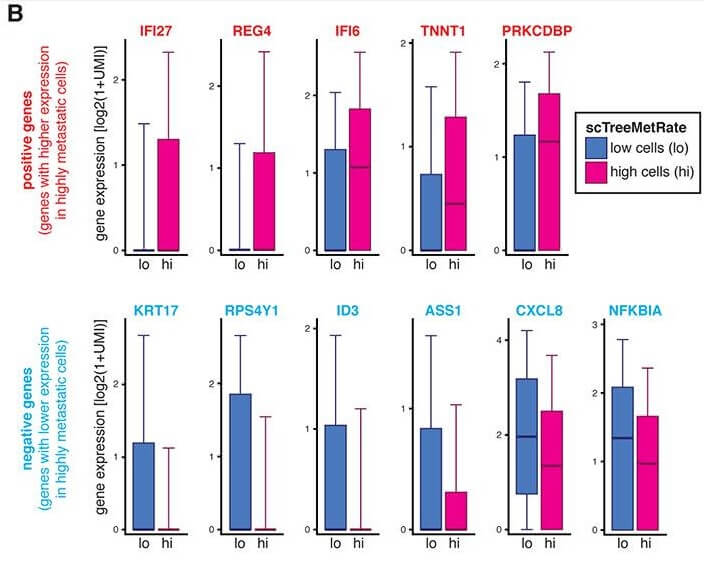 Image Courtesy: Quinn et. al, 2021
Image Courtesy: Quinn et. al, 2021
The metastatic state tended to be stable over the growth of the tumor. However, in a small number of cases, a tumor grew from a low metastatic to a high metastatic potential. This suggests that metastatic states can be stably inherited but are capable of evolving in vivo.
In summary, using the phylogenetic trees generated by the ‘molecular flight recorder’ technology, the researchers were able to follow the routes, rates, and directionality of the metastasis. These findings will help us further learn about the mechanics of tumor metastasis and also predict their behavior.
Dr. Weissman concluded his talk by briefly talking about another study where they were able to monitor tumor progression from oncogenic mutation to aggressive metastatic cancer and understand the underlying molecular events that were driving it.
©www.geneonline.com All rights reserved. Collaborate with us: service@geneonlineasia.com









