未分類

FDA Issues Guidances Supporting Development of Gene Therapy Products
By Yulia Abushinova A recently published report from Allied Market Research estimates that the rapidly progressing field of gene therapy could reach a market size of $6,205.85 million by 2026. …
Read More
FDA Greenlights Tepezza as First Treatment for Thyroid Eye Disease
By Rajaneesh K. Gopinath, Ph.D. Yesterday, Dublin based Horizon Therapeutics received the early FDA authorization of its human monoclonal antibody, Tepezza (teprotumumab-trbw). With this, the drug became the first and only approved treatment for …
Read More
Ignavibacterium Cas9 Beats Thermostability Record, Functions at 100 Degree Celsius
By Rajaneesh K. Gopinath, Ph.D. Researchers isolate a novel heat resistant Cas9 nuclease from a hyperthermophilic bacterium that grows in a high-temperature environment. The CRISPR-Cas9 genome-editing tool has evolved rapidly …
Read More
Weekly in Asia 20190602-0608
Thermo Fisher Scientific’s NGS-based Companion Diagnostic Test Receives Reimbursement Coverage in Japan Tumor samples collected from non-small lung cancer (NSCLC) patients are often limited in quantity. Thermo Fisher Scientific’s Oncomine …
Read More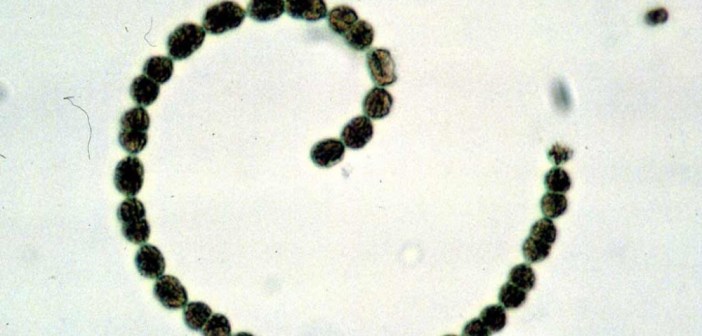
October 31, 2001: Genomic Sequence of a Specific Strain of Filamentous Cyanobacterium Anabaena sp. was Completed
The complete genomic sequence of a filamentous cyanobacterium, Anabaena sp. strain PCC 7120, was completed by an internatioanl team led by Takakazu Kaneko, Japanese microbiologist. The result was published in …
Read More
